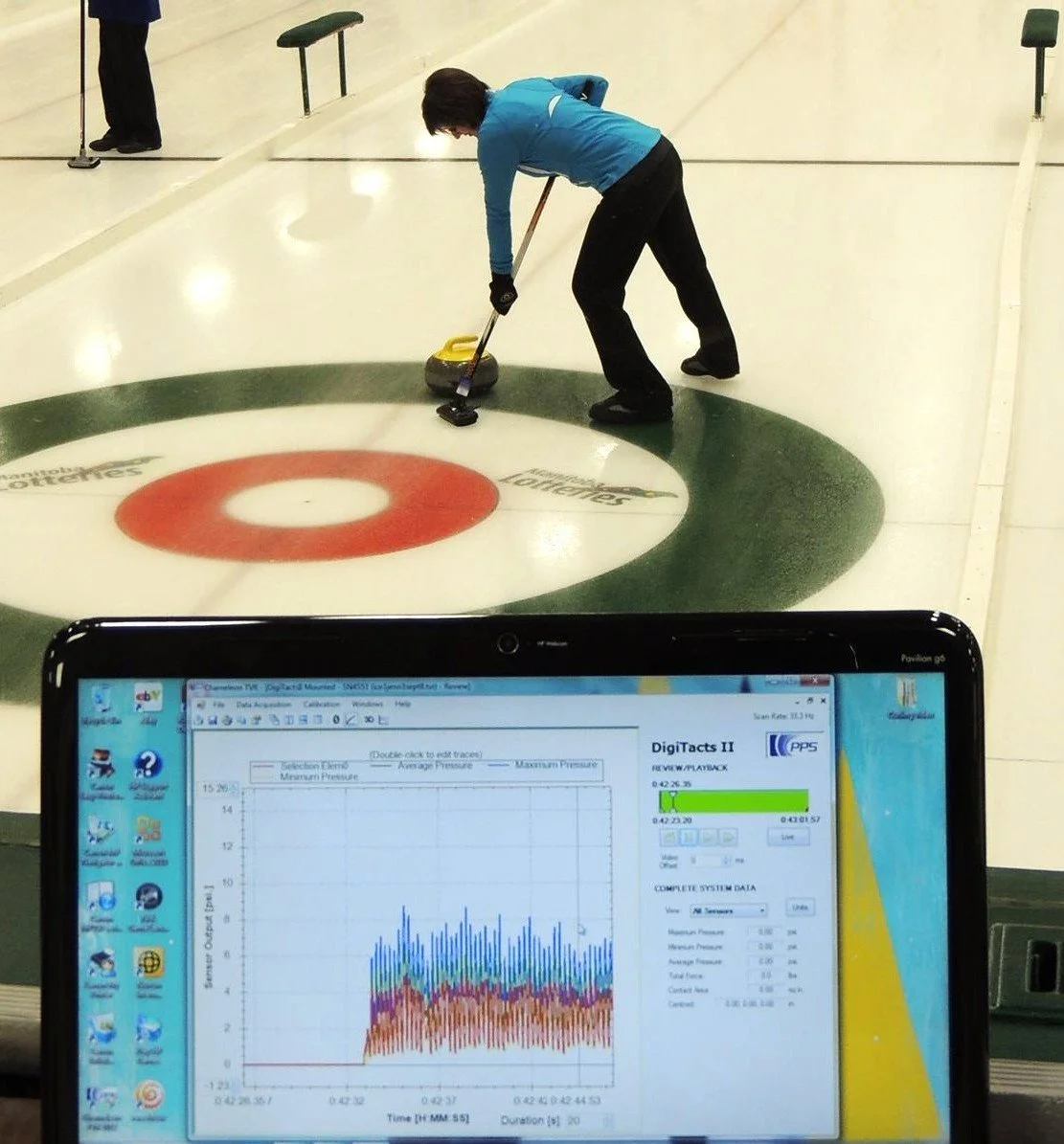
A purpose-built curling training brush developed with PPS enabled objective measurement of sweeping force, consistency, and fatigue during elite-level coaching sessions, providing performance insights that have supported teams competing at national, World Championship, and Olympic level.
Read MoreThis Heeluxe & PPS partnership shows how footwear expertise and tactile sensing technology combined to create SmartLast, transforming how footwear fit is evaluated and how design and comfort decisions are made.
Read MorePressure influences everything from the comfort of your glasses to the precision of a robotic gripper. Dr. Jae Son, founder of PPS, explains how tactile pressure mapping reveals how force is distributed, transforming invisible interactions into data that drives smarter, safer, and more comfortable designs.
Read MoreOver the past decade, the TactileGlove has evolved from a fragile prototype into a robust sensing system used in industry, healthcare, and sport. Each revision reflects lessons learned in materials, wiring, ergonomics, and connectivity - turning hard failures into lasting engineering insights.
Read MoreA fine mist spray may look simple, but achieving precision, consistency, and performance is a technical challenge. This article explores how sensor-based testing is transforming spray quality control in beauty and fragrance.
Read MoreCan ergonomic risk be measured with precision at scale? This Purdue-led study shows how TactileGloves and AI can transform how lifting tasks are assessed and safety decisions are made.
Read MoreMax Stancliffe’s study confirmed strong alignment between CFD predictions and measured pressure data. AeroStrip enabled accurate, non-intrusive pressure mapping on a Formula Student rear wing.
Read MoreTactile pressure sensing is transforming how designers approach fit, comfort, and performance. By providing real-time data on how garments interact with the body, this technology is reshaping everything from lingerie to medical wear, ensuring precision fit and lasting comfort.
Read MoreCapacitive tactile sensors provide high sensitivity, fast response, and durability for precise pressure measurement. This article explores their advantages, limitations, and how design improvements help overcome challenges.
Read MorePressure mapping is the measurement and visualization of pressure and its distribution when two surfaces come into contact. The measurement is usually preformed by a high-tech, thin, flexible and semiconductor sensor.
Read MoreTactile sensing technology, like FingerTPS™, is transforming inclusive product design by providing real-time data on fine motor skills, enabling more accessible and user-friendly solutions for diverse abilities.
Read MoreExplore the unique properties and applications of four key materials used in capacitive tactile sensors: conductive cloth, Lycra, Kapton, and hybrid designs. Learn how these materials enable innovations in wearable electronics, robotics, and industrial systems by balancing flexibility, durability, and sensitivity to meet specific needs
Read MoreCapacitive tactile sensing combines precise force measurement with detailed pressure distribution mapping using advanced materials and capacitance principles. This innovative technology enables thin, stretchable sensors ideal for diverse applications, from tactile arrays to conformable designs.
Read MoreTactile sensors are revolutionizing medical technology by enabling minimally invasive surgical tools that restore the sense of touch, enhancing catheter placement accuracy, and improving prosthetic limb functionality.
Read More